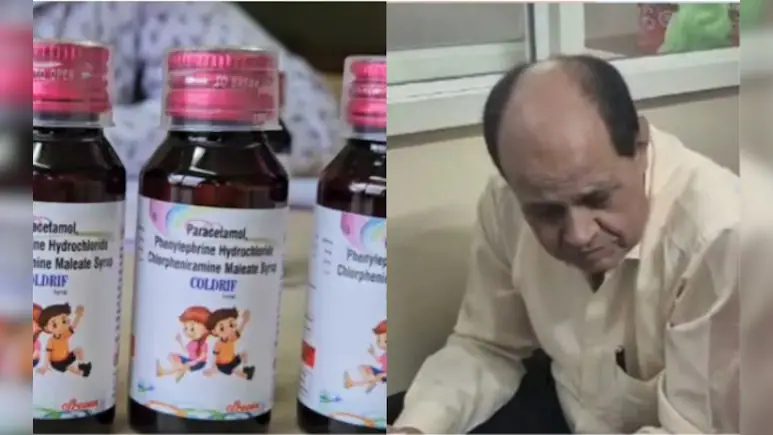
In a dramatic escalation of the ongoing cough syrup tragedy, authorities in Madhya Pradesh have arrested a doctor accused of prescribing a contaminated medication that allegedly led to the deaths of several children. The state government has also initiated legal action against Srisan Pharmaceuticals, the company that manufactured the contentious syrup.
The Arrest and Accusations
The arrested individual, Dr. Praveen Soni, is a pediatrician practicing in Chhindwara district. He had prescribed the Coldrif cough syrup, which preliminary investigations suggest was contaminated with diethylene glycol (DEG) — a toxic substance known to cause acute kidney injury. Samples of the drug reportedly contained approximately 48.6 percent DEG, triggering concerns over a mass poisoning event.
Most of the affected children were treated initially at Dr. Soni’s clinic in Parasia. They reportedly started with common cold or cough symptoms, but subsequently developed alarming signs such as reduced urine output, accelerated kidney dysfunction, and in many cases, death.
Government Action and Company Prosecution
Acting swiftly, the Madhya Pradesh government not only banned the sale of Coldrif syrup statewide but also filed a case against Srisan Pharmaceuticals, which is based in Kanchipuram, Tamil Nadu. The state accused the company of manufacturing a product that failed quality standards and caused fatalities.
In response to the ban, Srisan’s entire line of related medicines was also ordered to be withheld pending further investigation. Officials have asked that all existing stocks be frozen to prevent further distribution.
Regional Divergence in Responses
While Madhya Pradesh’s probe continues, states such as Tamil Nadu moved fast. Within 24 hours of receiving samples, Tamil Nadu’s Drug Control department declared that the batch in question was “Not of Standard Quality.” The state government immediately ordered a ban on distribution of that batch and alerted retail and wholesale drug inspectors.
Meanwhile, calls for tighter oversight and better regulation of pediatric drug manufacturing are intensifying across India, especially in the wake of multiple child fatalities linked to suspected syrup contamination.
While investigations are still underway, this move reflects the gravity with which the state is treating the crisis. The real measure of justice will be strict accountability—both at the individual and corporate levels—and robust reforms to prevent future tragedies.

Leave a Reply